|
12/2/2023 0 Comments Negative entropy  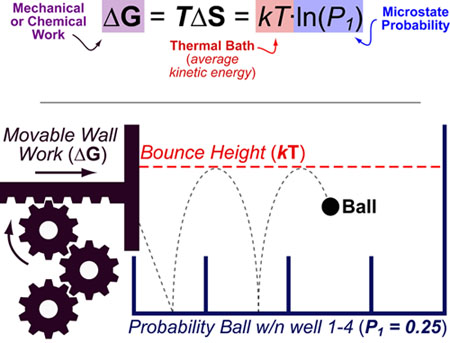
If you recall, we can express the total entropy change (the one the second law ”cares” about) as ΔStotal = ΔSsystem + ΔSsurroundings. And since we know that ΔHsystem = -ΔHsurroundings, (that is: the energy lost by the system = minus (-) the energy gained by the surroundings) we can express the entropy change of the surroundings in terms of measurable variables for the system. Mathematically we can express this as ΔSsurroundings = ΔHsurroundings/T. Since we know how much energy is added to the surroundings we can calculate the entropy change that this energy produces. Entropy is measured in units of J/K – that is energy/temperature. We can relate this thermal energy release to the entropy change of the surroundings. That is 6 kJ of thermal energy are released into the surroundings for every mole of water that freezes. The thermal energy change for the system, ΔHfreezing, is about -6 kJ/mole. Let us think about water freezing again: we can measure the enthalpy change for this process. Fortunately there is a reasonably simple way to do this. However, it is almost always easier to look at the system than it is to look at the surroundings (after all we define the system as that part of the universe we are studying.) It would therefor be much more convenient to use criteria for change that refer only to the system. This idea, that we must consider changes in entropy for both the system and its surroundings when we are predicting which way a change will occur (that is, in which direction a process is thermodynamically favorable) is an important one. The Second Law is preserved (yet again), but only when we actively embrace systems thinking. The increase in entropy in the surroundings is greater than the decrease in entropy of the ice! When we consider both system and surroundings, the change in entropy (ΔS) is positive. 
The reason that the freezing of water does not violate the second law is because even though the system (ice) becomes more ordered and has lower entropy, the energy that is released to the surroundings makes those molecules move faster, which leads to an increase in the entropy of the surroundings. Now we can see the solution to our thermodynamic problem. That is, the freezing of water is an exothermic process. Because energy is conserved, this energy must be released to the surroundings as thermal (kinetic) energy. As we have seen previously, the formation of stabilizing interactions lowers the potential energy of the system. 
Consider when water freezes, the water molecules form stable interactions (hydrogen bonds). However for the universe as a whole (or more easily defined, the system and its surroundings) total entropy must increase. How can we resolve this seeming paradox? The answer lies in the fact that for any system the entropy may indeed decrease - water freezing is an example of this phenomenon. The problem with this is that we are all well aware of changes where the entropy apparently decreases. The Second Law of Thermodynamics tells us that for every change that occurs, the entropy of the universe must increase. How can it be that a change in which the entropy of the system decreases (for example freezing of ice) can occur? Are we forced to conclude that things we know to happen are impossible according to the Second Law of Thermodynamics? We can calculate how entropies change for materials as they go from gas to liquid to solid, and as we have predicted they decrease. How do we think about entropy in these systems? Doesn’t a substance become more ordered as we move it from gas to liquid to solid? Clearly the entropy of a solid is lower than that of a liquid (and the entropy of a liquid is lower than that of a gas). Now let us return to the situation with solids, liquids, and gases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
